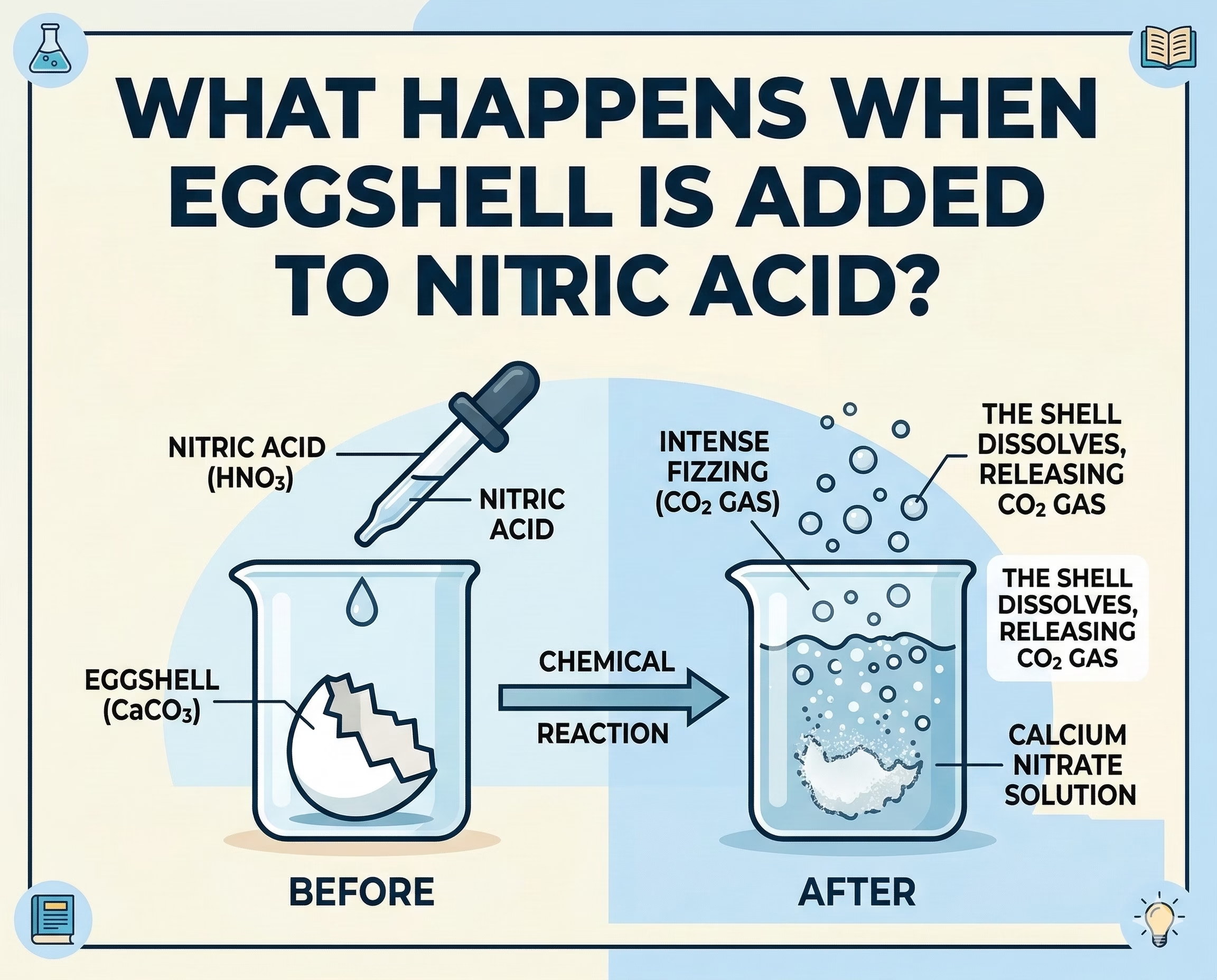
Chemical reactions between everyday materials and laboratory chemicals often reveal fascinating insights into chemistry. One interesting example is the reaction that occurs when an eggshell is placed in nitric acid. Although eggshells appear simple, they are made of chemical compounds that can react strongly with acids.
Understanding this reaction helps explain basic principles of chemistry such as acid reactions, calcium compounds, and gas formation. It is also a common experiment used in classrooms to demonstrate how acids react with carbonate materials.
In this article, we will explore what happens when eggshell is added to nitric acid, the chemical reaction involved, the products formed, the science behind the reaction, and the safety considerations associated with this experiment.
What Is an Eggshell Made Of?
To understand the reaction, it is important to know the composition of an eggshell.
Eggshells are primarily made of calcium carbonate (CaCO₃). In fact, about 94–97 percent of an eggshell is calcium carbonate, which is the same compound found in materials like chalk, limestone, and marble.
The remaining portion of the shell contains:
- small amounts of magnesium carbonate
- calcium phosphate
- organic proteins that provide structure
The outer shell protects the egg and gives it strength, but chemically it behaves similarly to other carbonate minerals when exposed to acids.
Because calcium carbonate reacts with acids, eggshells will react when placed in nitric acid, which is a strong mineral acid.
What Is Nitric Acid?
Nitric acid is a strong inorganic acid with the chemical formula HNO₃. It is widely used in industry and laboratories for several purposes, including:
- manufacturing fertilizers
- producing explosives
- metal processing
- laboratory experiments
Nitric acid is known for being highly reactive with many substances, especially carbonates and metals.
When nitric acid reacts with calcium carbonate, a chemical reaction occurs that produces new compounds and releases gas.
The Chemical Reaction Between Eggshell and Nitric Acid
When an eggshell is placed in nitric acid, the calcium carbonate reacts with the acid to form new substances.
The chemical reaction can be written as:
CaCO₃ + 2HNO₃ → Ca(NO₃)₂ + CO₂ + H₂O
In simple terms, the reaction produces:
- calcium nitrate
- carbon dioxide gas
- water
The carbon dioxide gas escapes as bubbles, which is why the reaction often appears as fizzing or bubbling when the eggshell touches the acid.
What You Observe During the Reaction
When an eggshell is added to nitric acid, several visible changes occur.
First, the eggshell begins to dissolve. This happens because the acid breaks down the calcium carbonate that forms the shell.
Second, bubbles appear around the shell. These bubbles are carbon dioxide gas being released as the reaction occurs.
Third, over time the shell becomes thinner and eventually disappears completely if enough acid is present.
These observations are typical for acid-carbonate reactions.
Why Carbon Dioxide Gas Is Released
Carbon dioxide is released during the reaction because of the breakdown of the carbonate ion in calcium carbonate.
Calcium carbonate contains the carbonate ion (CO₃²⁻). When an acid reacts with carbonate compounds, the carbonate ion reacts with hydrogen ions from the acid to produce carbon dioxide and water.
This is a standard chemical reaction that occurs whenever acids react with carbonates.
The gas produced during this reaction is the same gas released when baking soda reacts with vinegar.
Formation of Calcium Nitrate
Another important product of the reaction is calcium nitrate, which is formed when calcium from the eggshell reacts with nitric acid.
Calcium nitrate has the chemical formula Ca(NO₃)₂ and is highly soluble in water.
This compound is widely used in agriculture as a fertilizer because it provides plants with both calcium and nitrogen nutrients.
During the reaction, the calcium from the shell combines with nitrate ions from the acid to produce calcium nitrate dissolved in the solution.
The Role of Acid Strength
Nitric acid is considered a strong acid, meaning it completely dissociates in water to release hydrogen ions.
These hydrogen ions are responsible for reacting with calcium carbonate.
If a weaker acid were used instead, the reaction would still occur but at a slower rate.
For example:
- vinegar (acetic acid) reacts slowly with eggshell
- nitric acid reacts much faster
This difference happens because strong acids provide more hydrogen ions for the reaction.
Comparison With Other Acid Reactions
The reaction between eggshell and nitric acid is similar to reactions involving other acids and carbonate materials.
For example:
Eggshell + hydrochloric acid
CaCO₃ + 2HCl → CaCl₂ + CO₂ + H₂O
Eggshell + acetic acid (vinegar)
CaCO₃ + CH₃COOH → calcium acetate + CO₂ + H₂O
In all these reactions, carbon dioxide gas is produced.
The main difference is the type of salt formed after the reaction.
Applications of This Reaction
The reaction between calcium carbonate and acids has practical uses in several fields.
In geology, scientists use acid reactions to identify carbonate rocks such as limestone.
In agriculture, calcium compounds are used to adjust soil acidity.
In laboratories and classrooms, this reaction helps students understand concepts such as:
- acid-base reactions
- gas formation
- chemical equations
- carbonate chemistry
Because eggshells are easily available, they are often used in educational experiments.
Eggshell Dissolution Experiment
A simple experiment can demonstrate this reaction.
Materials needed:
- eggshell
- diluted nitric acid
- glass container
- safety equipment
Procedure:
First place a small piece of eggshell into a container.
Next add a small amount of diluted nitric acid.
Almost immediately, bubbles will form around the shell. These bubbles indicate the release of carbon dioxide gas.
Over time, the shell will gradually dissolve as the reaction continues.
This experiment clearly demonstrates how acids react with carbonate materials.
Safety Considerations
Although the reaction is simple, nitric acid is a dangerous chemical and must be handled carefully.
Nitric acid is highly corrosive and can cause severe burns if it touches skin.
Safety precautions include:
- wearing gloves
- wearing safety goggles
- working in a well ventilated area
- using diluted acid when possible
In most classroom experiments, weak acids such as vinegar are used instead of nitric acid to reduce risks.
Proper laboratory safety procedures should always be followed.
Why This Reaction Is Important in Chemistry
The reaction between eggshell and nitric acid demonstrates several important chemistry concepts.
These include:
acid reactions with carbonate compounds
gas formation during chemical reactions
dissolution of solid materials in acid solutions
formation of salts in chemical reactions
Because the reaction produces visible bubbles and dissolves the shell, it provides a clear example of chemical change.
Students often study this reaction while learning about acid-base chemistry.
Environmental and Natural Reactions
Similar reactions occur in nature as well.
For example, acid rain can slowly dissolve limestone and marble structures because these materials also contain calcium carbonate.
Over time, acidic rainwater reacts with carbonate minerals and gradually erodes them.
This is why many historic buildings made from limestone show signs of damage in polluted environments.
The eggshell experiment is a small-scale version of this natural chemical process.